This article will examine why air should be purged, and how it should be done.
Efficiency Down, Maintenance Up
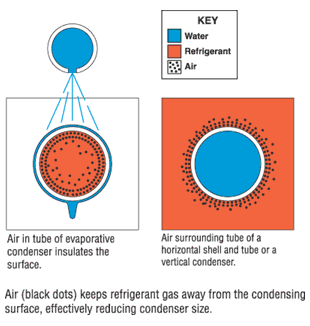
Air in the condenser will raise head pressure, mainly due to its insulating properties. Air molecules in the gas from the compressor will be blown to the quiet end of the condenser. This air accumulates on the heat transfer surfaces. (See Figure 1.)When condenser surfaces are insulated with air, the effective condenser size is reduced. This size reduction is offset by increasing the temperature and pressure of the refrigerant gas, which is an expensive luxury.
For each 4 pounds of excess head pressure caused by air, there is an increase of compressor power costs by 2 percent and reduced compressor capacity by 1 percent. Losses caused by reduced capacity may far exceed the extra costs while operating the compressor. More cooling water will improve condenser performance, but cooling water is also expensive.
Excess head pressure also puts more strain on bearings and drive motors. Belt life is shortened and gasket seals are ruptured.
Increased pressure leads to increased temperature, which shortens the life of compressor valves and promotes the breakdown of lubricating oil. Increased head pressure increases the likelihood of premature gasket failures.
Some so-called “ammonia explosions” have actually been traced to the accumulation of noncondensable hydrogen.
How Air Gets Inside
Air can enter any refrigeration system:To determine the amount of air in a refrigeration system, check the condenser pressure and temperature of the refrigerant leaving the condenser against the data in a temperature-pressure chart. If, for example, your ammonia temperature is 85 degrees F, the theoretical condenser pressure should be 151.8 psig.
If your gauge reads 171 psig, you have 20-psi excess pressure that is increasing power costs 10 percent and reducing compressor capacity by 5 percent.
If you do not have digital readouts or a liquid line mercury well, reasonably good readings can be obtained by strapping the bulb of an immersion thermometer to the liquid line. Cover the bulb with about 1 inch of Presstite Permagum, then insulate it with heavy cloth. In three to four minutes, it should be possible to take a reading accurate to within one-half to three-fourths of a degree.
Note: Air is not the only cause of excessive condenser pressure. A condenser that is too small or a condenser with fouled and scaled tubes will give high pressure readings without any air in the system. Air, however, is by far the most likely cause of excess condenser pressure. The air must be purged before the head pressure can be reduced to the proper level.
Proper Purge Procedures
Manual purging is too expensive and troublesome, except for very small systems. It doesn’t take a large percentage of air to cause a noticeable increase in high-side pressure. Manual purging at the condenser or receiver will discharge much more refrigerant than air into the atmosphere.Worse yet, as the air is purged from the system, even larger quantities of refrigerant must be wasted to get rid of the remaining air.
Besides wasting refrigerant, manual purging:

Refrigerated purging: Table 1 illustrates the principles of refrigerant purging and why it is needed. The table is based on an ammonia system. In lines 1-4, the temperature is held constant while the amount of air varies.
Note how the total pressure (high-side pressure) rises (columns E and F). Even when there is enough air to significantly raise the high-side pressure, the gas mixture is still mostly refrigerant (columns I and J).
In lines 5-10, the total pressure is held constant. As the purger is chilled, the refrigerant pressure drops. The balance of the pressure is due to the air, so this means that the concentration of air inside the purger is increasing. (See Figure 2.)
Line 2 represents a moderately low amount of air in the system. Achieving this condition by manual blowdown means that 28 pounds of ammonia is lost for every pound of air removed. By keeping the same total pressure as line 2, but cooling the gas to 0 degrees as shown in line 8, only 0.13 pounds of ammonia is lost when purging 1 pound of air. This means the refrigerant purge is 215 times as effective.

Similar gains will be seen in an R-134a system. (See Table 2.) Note, however, that obtaining low weight ratios of refrigerant gas to air may require lower temperatures than for the ammonia system.
The pressures and required purger temperatures will vary with other refrigerants, but the principles remain the same.

Where To Make Purge Connections
A refrigerated purger does not have magic fingers that can reach into a refrigeration system and find air. It is a device that will separate air from refrigerant gas in a purge system. Therefore, purge point connections must be located at places where air will collect.Refrigerant gas enters a condenser at high velocity. By the time the gas reaches the far (and cool) end of the condenser, its velocity is practically zero.
This is where the air accumulates and therefore where the purge point connection should be made. Similarly, the purge point connection at the receiver should be made at a point furthest from the liquid inlet. (Purge point connection locations are based on thousands of successful purge installations.)
Be prepared to purge from the condensers and receivers. Air will migrate from the condenser to the receiver and back again, depending on the load and plant conditions.
Air will remain in the condensers when the receiver liquid temperature is higher than condenser liquid temperature. This can happen when:
Conversely, air will migrate to the receiver when the condenser liquid temperature is higher than the receiver temperature. This can happen when:
The material was prepared by Armstrong International and is based on its publication, “The Guide to Refrigerated Purging,” Bulletin 702-D. Armstrong can be contacted at www.armstrong-intl.com.
Publication date: 06/02/2003

Report Abusive Comment