Making Sense of Refrigerant Blends
Refrigerant blends have been developed for existing as well as for new equipment. Their properties make them comparable alternatives to previously used substances.
Although the number of blends in the market has thankfully dropped off, the range of offerings is still very extensive.
There are two categories: transitional (service) blends and chlorine-free blends.
Blends in the field
Most transitional or service blends contain HCFC-22 as the main refrigerant. They are primarily service refrigerants for existing equipment that once used R-12, -502, or other CFCs. Correspond-ing products are offered by various manufacturers. Practical experiences covering the necessary steps of conversion are well known.Chlorine-free HFC blends are long-term substitutes for CFCs and HCFCs. HFCs 404A and 507A are already being used to a great extent.
Two- and three-component blends have a long history in the refrigeration trade. There is a difference between the so-called “azeotropes” (e.g., R-502) with thermodynamic properties similar to single-substance refrigerants, and “zeotropes” with gliding phase changes. The development of zeotropes mainly concentrated on special applications in low-temperature and heat pump systems.
A somewhat more common practice earlier was the mixing of R-12 and -22 in order to improve the oil’s return flow. R-22 was also added to R-12 systems to improve performance, as well as the addition of hydrocarbons in the extra-low-temperature range. The background for this was to provide better oil transport in the colder parts of the system, but also (with R-22/R-12) to reduce the discharge gas temperature with higher-pressure ratios.
This possibility of a specific “formulation” of certain characteristics was the main starting point for the development of a new generation of blends.
Looking for quick answers on air conditioning, heating and refrigeration topics? Try Ask ACHR NEWS, our new smart AI search tool. Ask ACHR NEWS
No directly comparable substitutes for R-502 and -22 are available from the chlorine-free, single-substance alternative refrigerants.
If flammability and toxicology are issues, and COP, pressure, and temperature conditions are to be comparable, the only substitutes that remain for the long term (in addition to R-134a) are blends.
Substitutes
Substitutes for R-502 have first priority, as they are needed in large quantities and are already affected by phaseout regulations in many countries.A zeotropic blend (such as R-502) behaves as a single-substance refrigerant with regards to evaporation or condensing processes. The phase change with zeotropic fluids occurs in a “gliding” form over a certain range of temperatures.
This temperature glide can be more or less pronounced. It is mainly dependent upon the boiling points and the percentage proportions of the individual components.
Certain supplementary definitions are also being used depending on the effective values. These terms include “near-azeotrope” or “semi-azeotrope” for a fairly small glide.
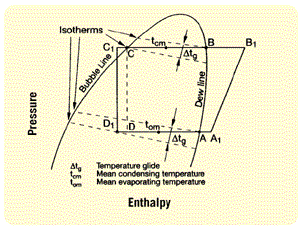
In practice, this means a small increase in temperature in the evaporation phase and a reduction during condensing. In other words, based on certain pressures, the resulting saturation temperatures differ in the liquid and vapor phases (Figure 1).
To enable a comparison with single-substance refrigerants, the evaporating and condensing temperatures have been mostly defined as mean values. As a consequence, the measured subcooling and superheating conditions (based on mean values) are unreal. The effective results — related to dew and bubble temperature — are less in each case.
These factors are very important when assessing the minimum superheat at the compressor inlet (usually 5 to 7 K) and the quality of the refrigerant after the liquid receiver.
With regard to a uniform and an easily comprehensible definition of the rated compressor capacity, there are international (EN12900) and U.S. (ARI540) standards to apply. Evaporating and condensing temperatures refer to saturated conditions.
Referring to Figure 1, the evaporating temperature is according to Point A, and the condensing temperature is according to Point B. Also in this case, the assessment of the effective superheating and subcooling temperatures will be simplified.
It must, however, be considered that the actual refrigeration capacity of the system can be higher than the rated compressor capacity. This is partly due to an effectively lower temperature at the evaporator inlet.
A further characteristic of zeotropic refrigerants is the potential concentration shift when leakage occurs. Refrigerant lost in the pure-gas and liquid phases is mainly noncritical. Leaks in the phase-change area (such as after the expansion valve, and within the evaporator and condenser-receiver) are considered more significant. It is, therefore, recommended that soldered and welded joints are used in these sections.
Extended investigations have, in the meantime, shown that the effect of leakage leads to less-serious changes in concentration than was previously thought. In any case, it is certain that the substances that are dealt with here cannot develop any flammable mixtures, either inside or outside the circuit. Essentially similar operating conditions and temperatures as before can only be obtained by supplementary charging with the original refrigerant in the case of a small temperature glide.
Practical Handling
Further conditions and recommendations concerning the practical handling of blends must also be considered.Renz is with Bitzer International of Sindelfin-gen, Germany. Information can be obtained through Delta Heat Transfer, 770-967-0030.
Publication date: 05/14/2001
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!




